Methane’s omnipresence is a testament to its transformative potential across industries, revolutionizing the world in which we live. This green energy not only generates power but also mitigates the impact of waste disposal on the environment. As an essential component of biogas, it emerges as a sustainable resource harnessed from organic waste. In the industrial landscape, methane acts as the cornerstone of chemical synthesis, paving the way for the creation of essential products and materials that shape modern society.īeyond its role in energy and manufacturing, methane’s versatility extends to agriculture.

Powering homes, businesses, and even vehicles, methane lights up our lives while diminishing the ecological burden. Embracing its clean-burning attributes, it takes center stage as a prized fuel source. Step beyond the natural realm, and methane reveals its versatility as a key player in numerous industries. Methane: Empowering Industries and Fueling Progress Striking the delicate balance between its contributions and challenges is a quest that scientists and environmentalists passionately pursue. On the other, it offers potential solutions for sustainable energy, serving as a clean-burning fuel that reduces carbon emissions. On one hand, its influence exacerbates the issue of global warming, intensifying climate change with its heat-trapping prowess. As a potent greenhouse gas, it dons the dual role of villain and hero in Earth’s atmosphere. Pockets of methane dance beneath icy surfaces on distant moons and planets, revealing the universal ubiquity of this simple hydrocarbon.īeyond its celestial charm, methane’s ecological impact is profound. Moreover, its occurrence extends beyond the planet’s crust. Found in vast reservoirs beneath the Earth’s surface, methane plays hide-and-seek within subterranean realms, waiting to be harnessed for various purposes. While methane may be inconspicuous to the human eye, it weaves its presence throughout the natural world. As the building block of life, methane is the cornerstone on which complex organic molecules emerge, crafting the foundation for all living organisms.Ībundance in Nature and Ecological Relevance Its chemical formula, CH4, encapsulates the harmony of carbon’s covalent bond with hydrogen, forming a symphony of atoms that constitute the backbone of organic compounds. Comprised of just one carbon atom and four hydrogen atoms, this humble gas boasts a molecular structure that captivates chemists and enthusiasts alike. Let’s dive into the depths of methane, its chemical formula CH4, and the fascinating role it plays in both nature and various industries.ĭefining Methane: Unraveling Nature’s Building BlockĪt its core, methane is the essence of simplicity. This colorless and odorless gas, composed of one carbon atom and four hydrogen atoms, wields a remarkable significance in the world of chemistry and beyond.
/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)
Methane, the unsung hero of the hydrocarbon family, takes the stage as the simplest and most fundamental organic compound. If you’re interested in chemistry or want to learn more about the chemical nature of methane, this article will pique your interest and help you gain a better understanding of how it holds up. In this course, we will go over step-by-step how to calculate the molar mass of its molecule, as well as its environmental and economic implications. In this article, we’ll go over the secrets of methane’s molar mass and how it functions in chemistry. This seemingly simple number has a significant impact on chemical reactions and plays an important role in determining the composition of various substances. The mole contains four grams of fat per gram. Methane, according to the chemical formula CH4, has a molar mass of 16.
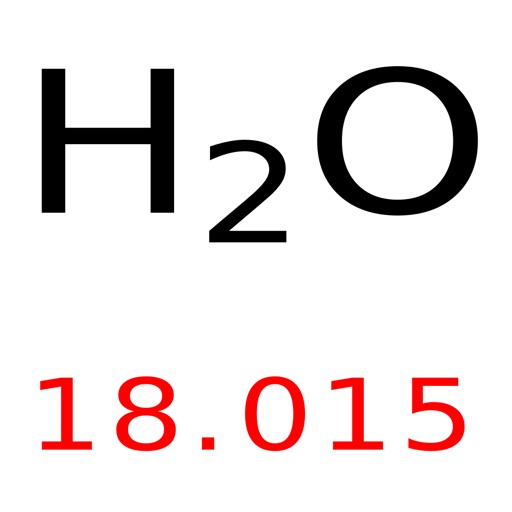
Methane is the most basic building block of organic chemistry, so what is the weight of this organic molecule? Let’s dive into the world of atoms and molecules, exploring the mystery behind the molar mass of methane.
